The priorities for research
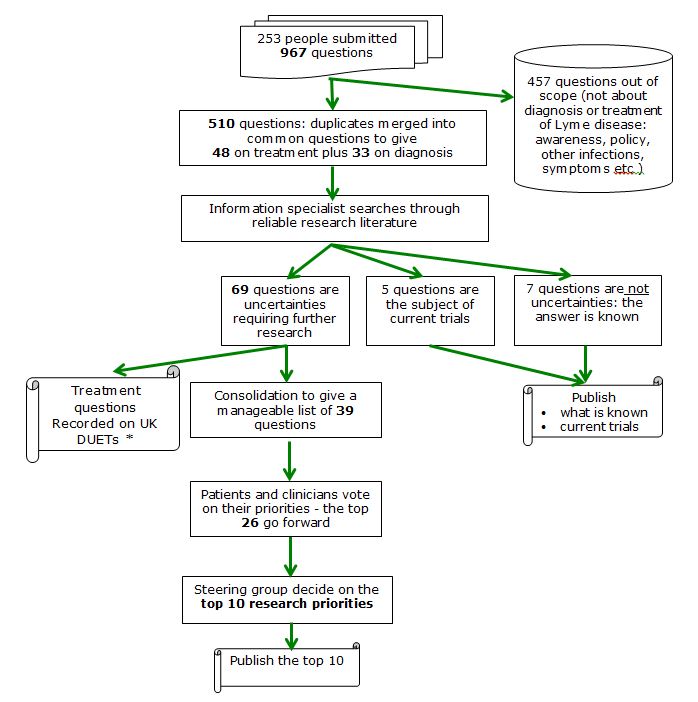
Through 2011-2012 LDA funded a process to discover some of the uncertainties in Lyme disease diagnosis and treatment. Via a James Lind Alliance Priority Setting Partnership patients and clinicians contributed to a survey which resulted in 81 potential uncertainties. Following a search of systematic reviews an independent information specialist confirmed that:
-
- 7 questions have known answers
- 5 are the subject of a current trial
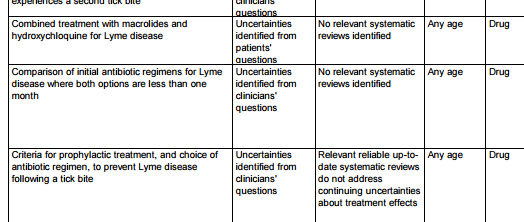
- 69 questions need further research
These 69 were consolidated and an on-line survey gave rise to the top 26. From these a group of doctors and patients decided on the top 10 shared priorities. The final workshop to decide on the top 10 was observed by the Department of Health and the HPA as witnesses to a rigorous, open process. Read about
- the full shortlist and the process
- the top 10 agreed by both clinicians and patients
The complete spreadsheet of uncertainties can be found on the JLA website.
These uncertainties are still outstanding, as verified by the NICE research recommendations published in April 2018.
Trials that were in progress
The following questions were also uncertainties. However, trials were underway at the time investigating these questions and so they were not included in the priority survey.
Details of the trials can be viewed on the WHO International Clinical Trials Registry Platform by entering in the search box the trial number provided below. For many of the trials there is an additional URL for ClinicalTrials.gov which provides more detail.
What is the best treatment when patients present with erythema migrans?
NCT01368341 (published) and NCT01163994 (published)
What is the best treatment for early Lyme neuroborreliosis in children and adults?
NCT01635530
Is intravenous antibiotic treatment more effective than oral treatment?
NCT01635530 and NCT01163994
Should chronic Lyme disease be treated with long term antibiotics?
EUCTR2009-010939-40-NL
Is an antibiotic gel on a tick bite an effective way of preventing Lyme disease?
EUCTR2011-000117-39-DE (published)
Known answers
The following are not uncertainties and the short answer to these questions is presented below.
Would homoeopathy be able to cure Lyme disease?
No. Trials in a number of conditions have shown that homoeopathy works no better than a placebo.
A Shang, K Huwiler-Muntener, L Nartey, P Juni, S Dorig, J A Sterne et al. Are the clinical effects of homoeopathy placebo effects? Comparative study of placebo-controlled trials of homoeopathy and allopathy, Lancet 2005, vol 366, pp 726-732
Does EM provide an ‘accurate’ clinical diagnosis of LD?
Yes it does.
Stanek G, Fingerle V, Hunfeld K, Jaulhac B, Kaiser R, Krause A, et al. Lyme borreliosis: clinical case definitions for diagnosis and management in Europe. Clinical Microbiology and Infection. 2011 Jan;17(1):69–79.
Is EM a more or less ‘accurate’ sign of LD in children?
Yes. Reference as above
How is EM defined?
See Stanek et al 2011 as above for description of European manifestations.
Should tests to exclude LD be carried out in patients who have a tick bite but don’t get EM?
In symptomatic patients, yes. EM is not always present
Stanek et al 2011
Tuerlinckx D, Glupczynski Y. Lyme neuroborreliosis in children. Expert review of anti-infective therapy. 2010 Apr;8(4):455–63.
Are there tests for other genospecies of Borreliae?
Yes; blood tests used in Europe include antigens from more than one species of Borrelia burgdorferi sl. However see other uncertainties regarding the genospecies in the UK.
Is there a test which can differentiate persistent active infection versus autoimmune response?
Yes, strictly speaking culture is a test which could be used. However it is not in routine diagnostic use. See related uncertainties on active infection.
James Lind Alliance Priority Sharing Partnership – how this PSP worked
The James Lind Alliance (JLA) is principally funded by the National Institute for Health Research and provides an infrastructure for patients and clinicians to work together to identify and confront the uncertainties about the effects of treatments. LDA is a JLA affiliate and provided the funding for the JLA Lyme Disease Priority Setting Partnership (PSP). The process is shown below, together with the number of questions at each stage.
* The UK Database of Uncertainties about the effects of Treatments (UK DUETs), absorbed into NICE and archived in January 2016.

 Printer Friendly
Printer Friendly